Overview
|
Our lab centers around the investigation of the neurocircuitry and neurobiology concerning the comorbidities of substance and alcohol addiction. Focusing on the manipulation of negative reinforcement, we are able to establish hyper-motivation for the excessive seeking and taking of drugs of abuse (including psychostimulants and opioids) and/or alcohol. We heavily employ extended drug access, self-administration paradigms as an animal model of addiction to mimic the transition from recreational drug use to compulsive dependency in humans. Furthermore, this self-administration model allows us to analyze and examine the neurobiological mechanisms that govern the compulsive behavior patterns of addiction and comorbid sleep dysfunctions that are associated with drug withdrawal and dependence. The current neurotransmitters and neuropeptides of interest include hypocretin/orexin, dynorphin, and norepinephrine.
Our main goal is to identify potential neurobiological targets for the pharmacological treatment of substance use disorders that can be used in combination with behavioral therapies in the clinical setting. |
Research Interests
Neurobiology underlying compulsive drug-taking
|
To study the neurobiology and neurocircuitry underlying addiction, we use a well-established rodent model of drug dependence in which rats are allowed to chronically self-administer (intravenous or vapor) drugs of abuse for extended periods. This paradigm has construct validity that mimics compulsive, dependent drug use in humans. In conjunction with other multidisciplinary techniques (pharmacology, adeno-associated viral (AAV) vectors, Designer Receptors Exclusively Activated by Designer Drugs (DREADD)-based chemogenetic tools, etc.), we can begin to dissect the neurocircuitry and molecular bases contributing to compulsive drug taking. We are particularly interested in how repeated cycles of drug withdrawal (physical and psychological) drive drug dependence. We are interested in how the neuropeptides (e.g., hypocretin/orexin), that are likely recruited during withdrawal, may contribute to compulive drug taking.
|
Sleep dysfunction and substance use disorders: the chicken or the egg?
|
Sleep dysfunction has been reported by nearly 90% of individuals with substance use disorders. Sleep disorders are also associated with both the development of substance use disorders and an increased likelihood to relapse following drug abstinence. Our lab seeks to characterize sleep dysfunction during drug abstinence (acute withdrawal and protracted abstinence) and discover the neurobiological-based changes associated with poor sleep in addiction. We use wireless radio telemetry recordings of electroencephalographic and electromyographic signals to determine sleep changes in rodents that are dependent on drugs of abuse. These data can help identify micro-architectural changes (e.g., sleep spindle frequency) to sleep structure that may lead to the discovery of relevant biomarkers of addiction and the propensity for future relapse.
|
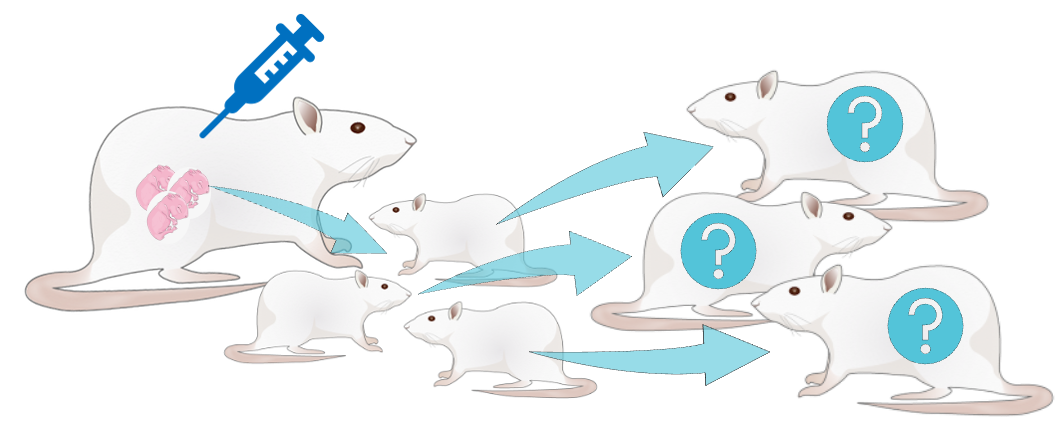
Effects of gestational opioids on offspring
|
In 2020, nearly 2.7 million people in the US had an opioid use disorder (OUD), dominated by prescribed opioids. Treatment strategies include prescription of an opioid agonist (methadone) or partial opioid agonist (buprenorphine) in medication-assisted treatment (MAT), including in women of reproductive age. Although MAT can benefit maternal opioid use disorder treatment, there has been a rise in incidence of infants born dependent on opioids due to gestational MAT exposure. Currently, gestational buprenorphine exposure is the primary cause of Neonatal Abstinence Syndrome (NAS) in Tennessee. Babies with NAS or neonatal opioid syndrome (NOWS) have smaller head circumferences, altered brain structures, and long-term learning disabilities. We seek to determine whether a lower dose MAT during gestation may mitigate some of the negative outcomes in offspring associated with higher doses of MAT in a rat model.
|